Dr. Diya Binoy Joseph - Research
Epithelial homeostasis and innate immune defenses in the urethra
Surfaces in the body that interact with the outside environment act as physical and biochemical barriers against environmental particulates and microorganisms. These surfaces are lined by specialized epithelial cells that intrinsically possess the capacity to interact with microorganisms. In addition, these epithelial barriers overlay a network of stromal and immune cells that mount responses to wounding and infection.
Although barrier defenses have evolved to handle the specific challenges confronting each organ system, there are certain features that are shared amongst these surfaces. Barrier surfaces have a robust regenerative capacity due to an abundant pool of stem/progenitor cells. These epithelial surfaces are the first line of defense against pathogens by the expression of antimicrobial defense genes. Another unifying feature is the presence of resident microbiota that co-exist with these epithelial structures and shape host defenses. These unifying features necessitate collaboration between scientists investigating barrier surfaces throughout the body. There is much to be discovered by comparing and contrasting epithelial barrier structures across the body and how they maintain homeostasis under the varying conditions they are exposed to.
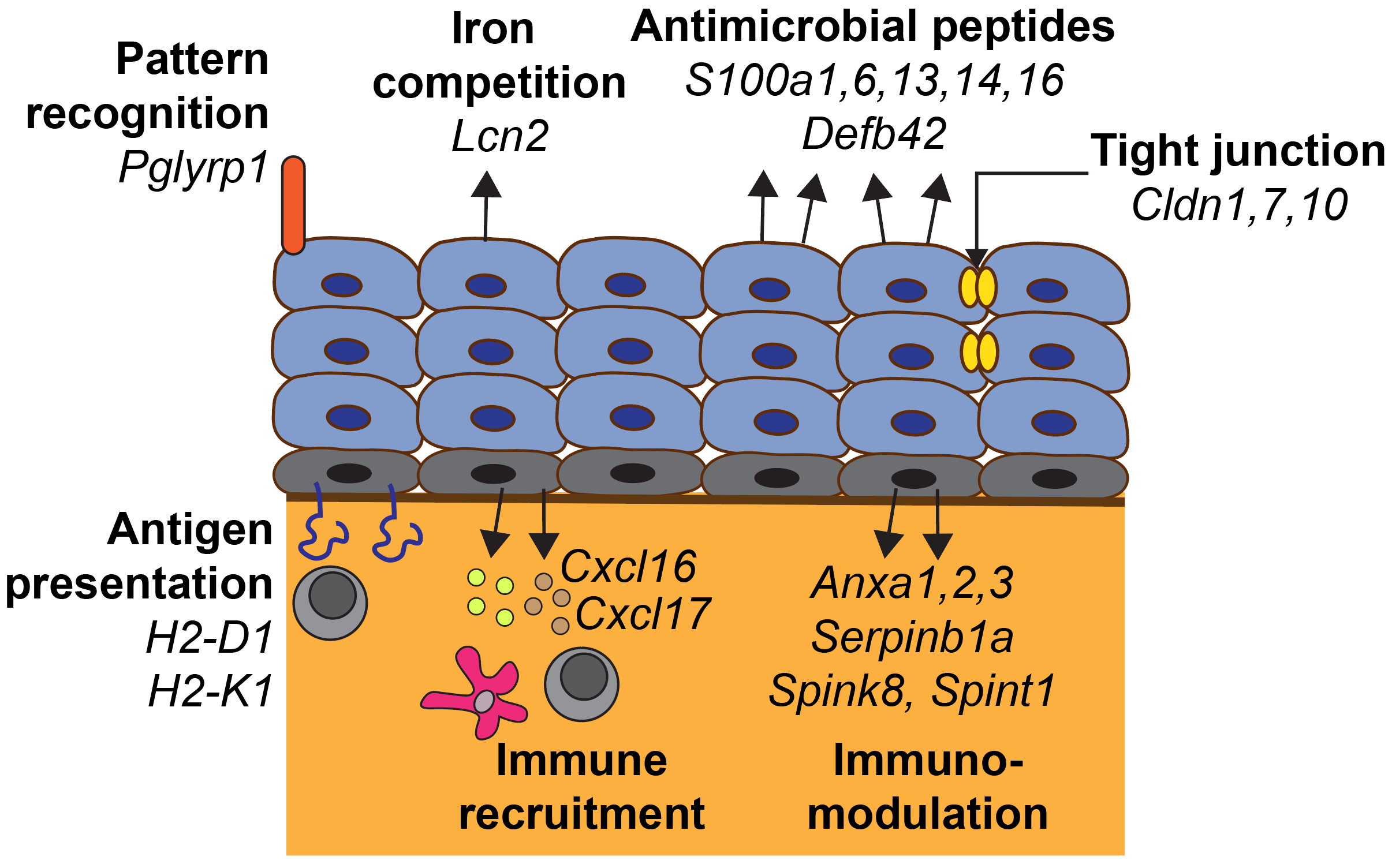
Defenses at the urethral barrier
One such barrier is the lining of the urethra, the tube connecting the bladder to the outside. Instead of acting as a passive lining, the epithelial cells of the urethra are highly specialized. Intercalated in the urethral epithelium are specialized neuroendocrine cells that secrete neurotransmitters upon activation. Recent work identified specialized club and hillock cells in the human urethral epithelium (Henry GH et al. Cell Reports 2018). Club cells represent a secretory epithelial cell type that expresses proteins involved in immune defense and xenobiotic metabolism. Interesting parallels exist between the protective mechanisms employed by the urethra and the lung, where club cells and neuroendocrine cells are well studied. The secretory profile of club cells matches that of other cells located near barriers including salivary gland cells and sweat gland cells.
In my postdoctoral work, I identified equivalents of human club cells in the urethral epithelium of the mouse (Joseph DB et al. Prostate 2020). My future work at inStem will focus on identifying the functional role of these urethral cells in protecting the bladder from infection. Urinary tract infections are highly prevalent and are initiated by pathogens ascending up the urethra to attach to the bladder epithelium. Using mouse models and human tissue culture, I will study:
1. How immune defenses in the urethra are initiated during development
2. The molecular mechanisms that determine the antimicrobial functions of urethral epithelial cells
3. The role of the urinary microbiome in shaping immune defenses in the urethra
Urethral cells express an arsenal of innate immune defenses that could limit bacterial infection of the bladder and in the case of males, the prostate. By understanding how immune defenses in the urethra are regulated, my goal is to limit infections before they spread to the bladder.


